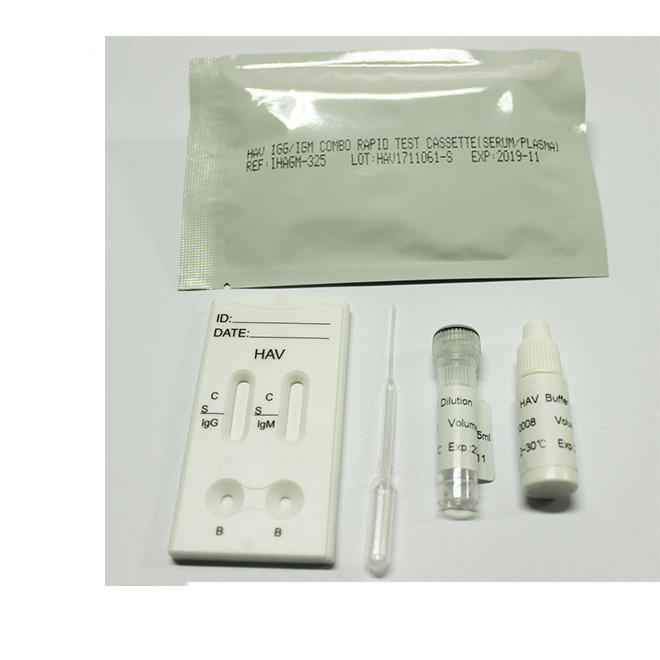
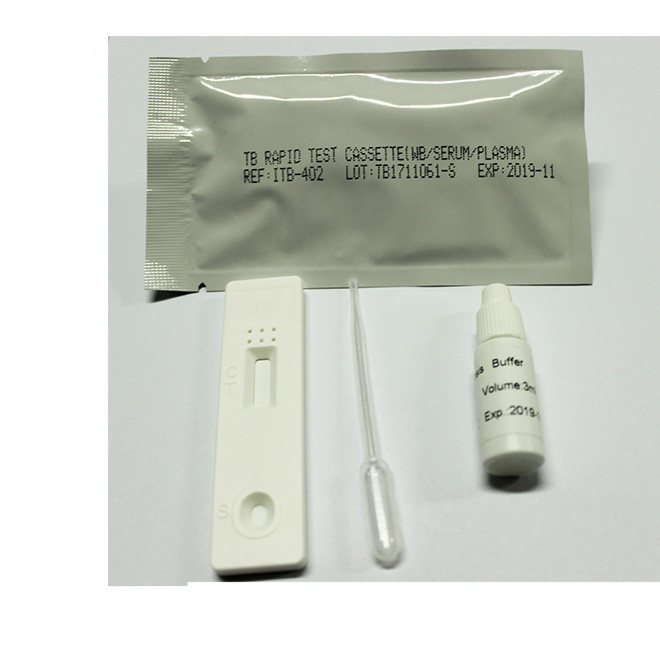
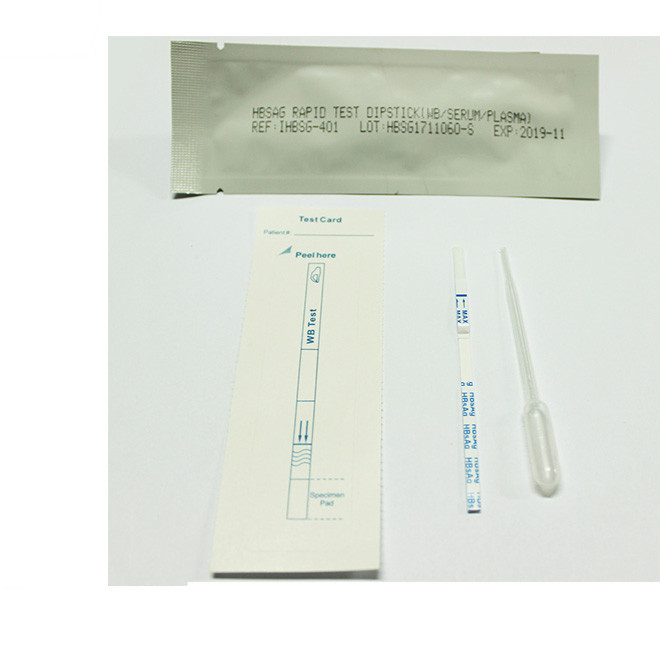
Quick Details
1. Fast.
2. High sensitivity and specificity.
3. Simple to use.
4. Accurate and reliable.
5. Ambient storage.
6. IgG and IgM combo. Screening of Typhoid current or past infection.
Packaging & Delivery
| Packaging detail:Standard export package Delivery detail:within 7-10 workdays after receipt of payment |
Specifications
AMRDT014 Reliable Typhoid Rapid Test Cassette
A rapid test for the qualitative detection of lgG and IgM antibodies to Salmonella typhi (S. typhi) in human whole blood, serum or plasma specimen. For professional in vitro diagnostic use only
[INTENDED USE]The Typhoid Rapid Test Cassette is a rapid chromatographic immunoassay for the simultaneousdetection and differentiation of IgG and IgM types of antibodies against Salmonella typhi (S. typhr) in human serum or plasma. It is intended to be used as a screening test as an aid in the diagnosis of infection with S. typhi. Any reactive specimen with the Typhoid rapid test cassette needs to be confirmed with alternative testing method.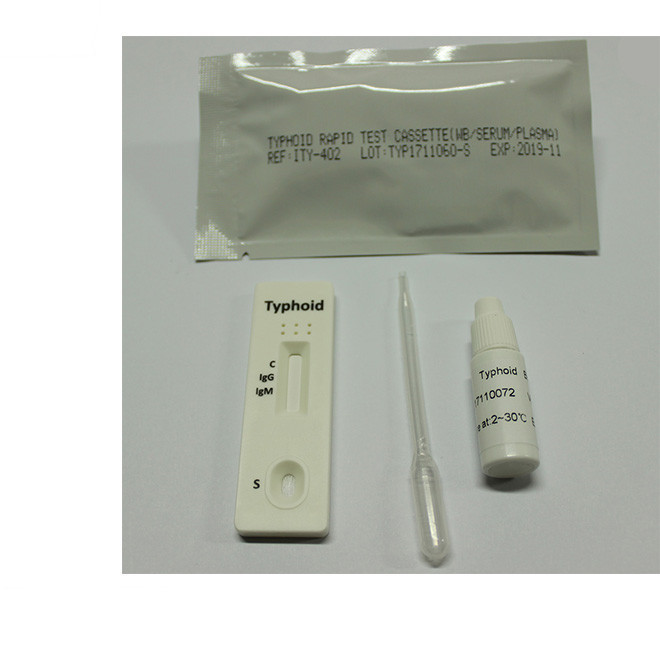
AMRDT014 Reliable Typhoid Rapid Test Cassette
1. Fast.
2. High sensitivity and specificity.
3. Simple to use.
4. Accurate and reliable.
5. Ambient storage.
6. IgG and IgM combo. Screening of Typhoid current or past infection.
|
Catalog No. |
AMRDT014 |
|
Product name |
Typhoid Rapid Test Cassette (Whole Blood/Serum/Plasma) |
|
Analyte |
IgG&IgM |
|
Test method |
Colloidal Gold |
|
Sample type |
WB/Serum/Plasma |
|
Sample volume |
1 drop |
|
Reading time |
15 mins |
|
Sensitivity |
IgM: 93.9% |
|
Specificity |
IgM: 99.0% |
|
Storage |
2~30℃ |
|
Shelf life |
24 months |
|
Qualification |
CE |
|
Format |
Cassette |
|
Package |
40T/kit |
AMRDT014 Reliable Typhoid Rapid Test Cassette
[SUMMARY]Typhoid fever is caused by S.加hi, a Gram-negative bacterium. World-wide an estimated 17 million cases and 600,000 associated deaths occur annually’. Patients who are infected with HIV are at significantly increased risk of clinical infection with S. typhi2. Evidence of h. pylori infection also presents an increase risk of acquiring typhoid fever. 1一%of patients become chronic carrier harboring S. typhi inthe gallbladder.The clinical diagnosis of typhoid fever depends on the isolation of S. typhi from blood, bone marrow or a specific anatomic lesion in the facilities that cannot afford to perform this complicated and time consuming procedure. Widal test (also referred as Weil Felix Test) is used to facilitate the diagnosis.however, many limitations lead to difficulties in the interpretation or the widal test.In contrast, the Typhoid Rapid Test Cassette is a simple and rapid laboratory test. The testsimultaneously detects and differentiates the IgG and the IgM antibodies to typhi specific antigens inwhole blood, serum or plasma thus aid in the determination of current or previous exposure the S. typhi.[PRINCIPLE]The Typhoid Rapid Test Cassette is a qualitative, membrane based immunoassay for the detection of antibodies (IgG and IgM) to Salmonella typhi (S. typhr) in human whole blood, serum or plasma. The diagnostic test cassette consists of two components: an IgG component and an IgM component. TheIgG line region is pre-coated with reagents for the detection of anti-S.typhi (IgG). The IgM line region is pre-coated with monoclonal anti-human IgM for detection of anti-S. typhi (IgM).During testing, specimen dispensed into the sample well of the test cassette binds with Typhoid conjugates impregnated in the reagent area, if the specimen contains anti-Typhoid antibodies. The immunocomplex thus formed m心rates by capillary action. If the present antibodies in specimen are ofIgG types, the immunocomplex is then captured by the pre-coated reagents on the membrane, forming a colored IgG line, indicating a S. typhi IgG positive test result. If the present antibodies in the specimen are of IgM type, the immunocomplex would be captured on the membrane by the pre-coated anti-human IgM antibody, forming a colored IgM line, indicating a S. typhi IgM positive test result.Absence of any T lines (IgM and IgG) indicates a negative result. A colored control line (C) should always appear in case of a positive or a negative result. Its absence indicates invalid test results[REAGENTS]The test contains mouse anti-human IgM, mouse anti-human IgG and Typhoid antigen. A goat antibody is employed in the control line system.
AM TEAM picture

AM Certificate

AM Medical cooperate with DHL,FEDEX,UPS,EMS,TNT,etc.International shipping company,make your goods arrive destination safely and quickly.